Background: Approximately 10% of adults with sickle cell disease (SCD) have diabetes mellitus (DM) (PMID 30714090). Glycemic control is challenging to evaluate in the setting of hemoglobinopathies, such as SCD, due to potentially inaccurate hemoglobin A1c (HbA1c) measurements (PMID 30897962). Fructosamine has been proposed as a marker of glycemic control in patients with hemoglobinopathies, although its use remains unvalidated in clinical studies (PMID 27457632). The ideal glycemic monitoring strategy in patients with SCD is unclear. Therefore, the objective of this study was to evaluate the performance of fructosamine and HbA1c in predicting glycemic burden in adults with SCD.
Methods: We analyzed 439 patients followed between Jan 2010 and Feb 2023 at the University of Illinois Sickle Cell Center. DM was defined by a diagnostic code in the electronic health record plus either prescription of an outpatient antidiabetic agent or an outpatient random serum glucose >200 mg/dL. Comparisons of clinical and laboratory variables by DM status were performed using the Wilcoxon rank sum, Pearson's chi-squared, and Fisher's exact test. Correlation analyses were conducted to evaluate the association of fructosamine and HbA1c with time-paired mean serum glucose concentrations, and of HbA1c with indirect bilirubin (I Bili), absolute reticulocyte counts (retic), and lactate dehydrogenase (LDH) concentration. Multivariate regression models for HbA1c used mean serum glucose concentration and either I Bili, retic, or LDH as covariates with an interaction term.
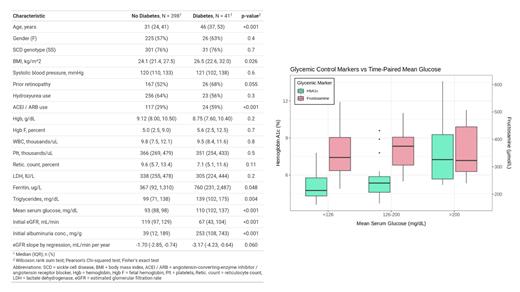
Results: The median age of the cohort at the time of enrollment was 32 years (interquartile range [IQR] 25-43 years), 57% were female, 76% were hemoglobin SS, and 46% were on hydroxyurea. 41 (9.3%) patients had DM (Table). SCD patients with DM were older, more frequently on angiotensin converting enzyme inhibitors or angiotensin receptor blockers, had higher serum triglyceride concentrations, lower baseline eGFR, and higher baseline urine albumin-to-creatinine ratios compared to those without DM (p ≤ 0.004).
In 39 patients with 157 observations, fructosamine concentration correlated poorly with 14-day time-paired mean glucose concentrations ( r s +0.02, p = 0.8; R2 0.01). In 19 patients with 176 observations, HbA1c significantly correlated with 30-day time-paired mean glucose concentrations ( r s +0.36, p < 0.001; R2 0.25). HbA1c progressively increased with higher glucose category (p < 0.001), while there was no association between fructosamine concentrations and glucose category (p = 0.5) (Figure).
Increased hemolysis was associated with decreased HbA1c (I Bili r s -0.44, p < 0.001; retic r s -0.33, p = 0.002; LDH r s -0.18, p = 0.2). After adjustment for hemolytic markers with interaction effects, HbA1c continued to predict 30-day time-paired mean glucose concentration (I Bili β HbA1c +20.8, p < 0.001; retic β HbA1c +24.1, p < 0.001; LDH β HbA1c +26.0, p = 0.002). The addition of these hemolytic and interaction terms generally improved the fit of the model (Unadjusted R2 0.24 versus adjusted for I Bili R2 0.42, retic R2 0.22, or LDH R2 0.55).
Conclusions: Our findings suggest that fructosamine concentration is a poor glycemic marker in adults with SCD. We observe that increased hemolytic markers are associated with decreased HbA1c, and the addition of a corrective hemolysis interaction term may improve the predictive ability of HbA1c for mean glucose concentration. Further studies are urgently needed to develop and validate glycemic monitoring strategies, such as continuous glucose monitoring, in this group of patients.
Disclosures
Layden:Bayer Health Care Pharmaceuticals: Consultancy. Gordeuk:Incyte: Research Funding; GBT/Pfizer: Consultancy, Research Funding; Novartis: Research Funding; CSL-Behring: Consultancy; Modus Therapeutics: Consultancy; Forma: Consultancy, Research Funding; Emmaus: Consultancy, Research Funding; Takeda: Consultancy. Saraf:Novartis: Consultancy, Other: Advisory board, Research Funding; GBT/Pfizer: Consultancy, Other: Advisory board, Research Funding, Speakers Bureau; Agios: Consultancy, Other: Advisory board; BEAM Therapeutics: Consultancy, Other: Advisory board; Forma Therapeutics: Consultancy, Other: Advisory board, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal